The first stage in the development of the action plan involved the definition of priority issues that were classified in two categories:
1. Regulatory Process
2. Technical Regulations
On July 1st, 2020, during the First Virtual Meeting of the Coalition, Principal Members presented their projects.
The Projects related activities developed by the Technical Secretariat were:
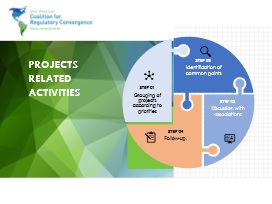
Every action plan was analyzed as to identify whether the cause was:
It was identified that the root cause of most regulatory problems experienced by the members begins with conducting inadequate regulatory processes.
Thus, the Technical Secretariat proposed to their Principal Members to hold tailor-made training in each country to further develop capacities among all related stakeholders on good regulatory practices, with the objective to ensure the creation or update of any technical regulation fulfills the local legal requirements as well as the countries treaties obligations.














